How Much Do You Know about COVID-19 Vaccines?
Dr. Melody Leung, Division of Life Science, explains the mechanisms behind COVID-19 vaccines
By Dr. Melody Leung, Division of Life Science, HKUST

The Coronavirus Disease 2019 (COVID-19) has been threatening the world for more than a year. We all wish the pandemic will be under control as soon as possible, with high expectations on vaccine development. At the end of 2020, the HKSAR Government announced the successful purchase of three COVID-19 vaccines, including the inactivated vaccine by Sinovac Biotech (Hong Kong) Limited, the ribonucleic acid (RNA) vaccine by Fosun Pharma / BioNTech, and the viral vector vaccine by AstraZeneca / Oxford University. People are very concerned about the mechanism of these vaccines and the potential side effects they may induce. How exactly do these vaccines protect us from viral invasion?
Once our body cells are infected by a virus, they will become the factory of the virus to duplicate more viruses based on instructions of its genetic material. Meanwhile, our immune system will be activated to trigger the actions of immune cells: some of them eliminate the virally infected body cells, some catch the viruses that wander around outside the cells, and some record the protein or other constituent features of the virus in order to help the immune system produce the corresponding weapons that target the virus. This way, when the virus invades again, the immune system will be able to identify the virus based on the records, quickly prepare the weapons, and organize a powerful attack to kill the virus within a short time.
Vaccines work by making use of the recording function of the immune system. The immune system memorizes and analyzes the viral features presented by the vaccines, so that we can get prepared in advance and fight the virus anytime when it attacks us. These viral features can be obtained from the dead or weakened virus, and the new generation of vaccines enables our body cells to produce these features with the viral genes, while the vaccines themselves are basically non-pathogenic.
The following will explain the mechanism and potential side effects of the three above-mentioned COVID-19 vaccines in detail.
The virus inactivation technology adopted in Sinovac’s vaccine is a conventional vaccine production method. Researchers utilize the kidney epithelial cells extracted from African green monkeys, also known as Vero cells, as a virus factory and, in the laboratory, conduct mass cultivation of the COVID-19 strain being isolated from the patients, before processing the virus using organic compounds to maintain the integrity of the spike protein (also known as S protein) on its outer layer as well as the other constituents, while disabling its replication at the same time. This is what “inactivation” means. However, once inactivated, the virus can hardly trigger our immune responses. And that’s why the vaccine will be mixed with aluminum-based immunoadjuvant in order to stimulate our immune system. Inactivation technology is relatively more sophisticated and easier to execute as a vaccine production technology. However, since a huge number of active viruses are processed during the vaccine production, a stringent laboratory management is extremely crucial. The efficacy of inactivation also needs to be strictly controlled to prevent the vaccinees from any unfortunate infection caused by active virus. In addition, although aluminum-based adjuvants, under normal circumstances, are not harmful to human body, some people are still worried that these adjuvants may induce adverse responses or side effects, especially on the nervous system.
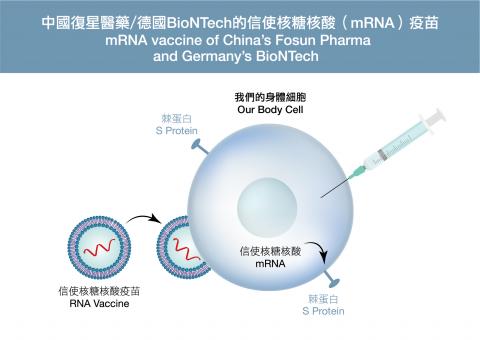
The messenger ribonucleic acid (mRNA) vaccine by China’s Fosun Pharma and Germany’s BioNTech is a next-generation of vaccine. The ribonucleic acid (RNA) of the COVID-19 spike protein is first reversely transcribed1 into a deoxyribonucleic acid (DNA) in the laboratory and then, with the use of equipment, transcribed2 in large quantities to the corresponding mRNA according to the DNA gene sequence. When the mRNA vaccine enters our body cells, it will eventually be translated3 into the viral spike proteins. Some of these spike proteins will appear on the surface of human cells or be released during cell apoptosis. The exposed spike proteins will stimulate our immune responses. As the viral spike protein is made by the cells of the vaccinees themselves, the immune system would be activated as if the cells are really infected by the virus. No adjuvant is required during the process. Moreover, there are natural enzymes in our body that degrade mRNA. So the mRNA vaccine will eventually disappear without leaving a trace. However, mRNA is very vulnerable. This is why the mRNA being used as vaccine will be protected in lipid nanoparticles and must be stored at extremely low temperature (-70oC). This stringent storage requirement has made transportation of vaccine very difficult and thus has deterred some developing countries from purchasing this vaccine. The side effects that may be induced by the mRNA vaccine are similar to those of other vaccines, including symptoms such as mild local skin redness and tingling, fever, muscle pain, and fatigue. And yet because this vaccine contains no viruses, it does not pose too much risk to human body.
The viral vector vaccine of Britain’s Oxford/ AstraZeneca Pharmaceutical is also a next-generation vaccine. The viral vector is like a container that delivers goods safely to the destination. The “container” being used in this vaccine is a modified chimpanzee adenovirus, and the “cargo” inside is the DNA being reversely transcribed from the RNA of the COVID-19 spike protein. When the viral vector enters the human body, the viral DNA inside will enter the cell nucleus, where it is transcribed into mRNA, and then translated into the COVID-19 spike protein. Like the mRNA vaccine by Fosun Pharma / BioNTech, the exposed spike protein will stimulate our entire immune response without adjuvants. The difference is that the DNA remaining in the nucleus is not easily degraded. Besides, the DNA carried by the viral vector is relatively more stable, and only requires a storage temperature of 2-8oC which makes it much easier to manage. The side effects are similar to those of mRNA vaccines, including tenderness at the injection site, fatigue, headache, muscle pain and fever.

It is worth noting that all the three vaccines rely on the unique features of the spike protein on the viral surface of the collected viruses that our immune system can recognize. When mutation occurs in the spike protein of the virus, there is a chance that the currently available vaccines may no longer protect us. As far as the above-mentioned three vaccines are concerned, the mRNA vaccine is the most direct and efficient among them in terms of production process, and is the first among them to enter the clinical trial stage in the race of COVID-19 vaccine development. According to the information of the World Health Organization as of 26 January 2021, 63 COVID-19 vaccines are currently in clinical trials, with an addition of 173 vaccines undergoing preclinical development. They include, in addition to the three types of vaccines being introduced in this article, the conventional protein subunit vaccines, as well as the mixed vaccines with added antigen presenting cells. It is believed that the development of vaccines will be more sophisticated in the future offering us better protection.
Remarks:
- Reverse transcription: The process of transcribing RNA into DNA
- Transcription: The process of copying DNA into RNA
- Translation: There are three kinds of RNA, in which mRNA carries the gene sequence of protein. The process of converting the mRNA sequence into protein is called translation